Vanadium Vanadium is a chemical element with symbol V and atomic number 23. Classified as a transition metal, Vanadium is a solid at room temperature. The atomic numbers of vanadium (V), Chromium (Cr), manganese (Mn) and iron (Fe) are respectively 23, 24, 25 and 26 Which one of these may be expected to have th.
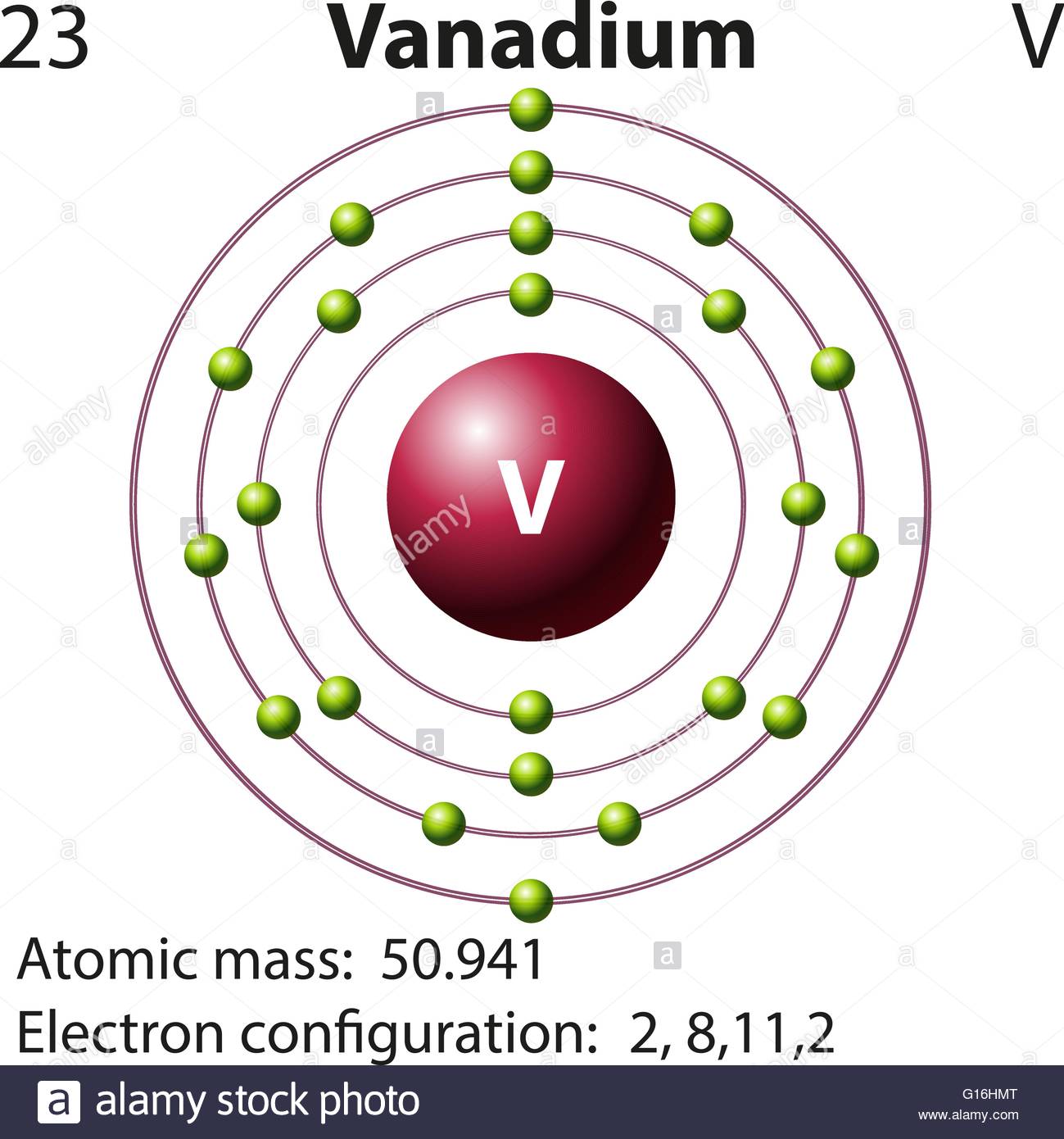
Atomic Number of Vanadium is 23.
Atomic Mass of Vanadium Atomic mass of Vanadium is 50.9415 u. Vanadium metal, sheet, strip, foil, bar, wire, and tubing have found use in high-temperature service, in the chemical industry, and in bonding other metals.Because the major commercial use of vanadium is in steel and cast iron, to which it lends ductility and shock resistance, most of the vanadium produced is used with iron as ferrovanadium (about 85 percent vanadium) in making vanadium steels.
Chemical symbol for Vanadium is V. Number of protons in Vanadium is 23. Atomic weight of Vanadium is 50.9415 u or g/mol. Melting point of Vanadium is 1890 °C and its the boiling point is 3380 °C.
» Boiling Point» Melting Point» Abundant» State at STP» Discovery YearVanadium Atomic Structure
About Vanadium

Vanadium is one of the strong metals with light grey color, which is very often used in alloys with steel to strengthen it. This element was named after one of the Scandinavian goddesses, and it was discovered in abundance in the Universe, but not so much on our planet. Living creatures need a bit of vanadium to support their life circle. This chemical element occurs naturally in some minerals. Vanadium is dominantly used in alloys with other metals, for producing various parts for machinery and automobiles. Alloys of this chemical element are also used in constructing nuclear reactors.
Properties of Vanadium Element
Vanadium Atom Model
| Atomic Number (Z) | 23 |
|---|---|
| Atomic Symbol | V |
| Group | 5 |
| Period | 4 |
| Atomic Weight | 50.9415 u |
| Density | 6.11 g/cm3 |
| Melting Point (K) | 2183 K |
| Melting Point (℃) | 1890 °C |
| Boiling Point (K) | 3680 K |
| Boiling Point (℃) | 3380 °C |
| Heat Capacity | 0.489 J/g · K |
| Abundance | 120 mg/kg |
| State at STP | Solid |
| Occurrence | Primordial |
| Description | Transition metal |
| Electronegativity (Pauling) χ | 1.63 |
| Ionization Energy (eV) | 6.7462 |
| Atomic Radius | 135pm |
| Covalent Radius | 125pm |
| Valence Electrons | 2 |
| Year of Discovery | 1801 |
| Discoverer | del Rio |
What is the Boiling Point of Vanadium?
Vanadium boiling point is 3380 °C. Boiling point of Vanadium in Kelvin is 3680 K.
Atomic Number Of Vanadium 51
What is the Melting Point of Vanadium?
Vanadium melting point is 1890 °C. Melting point of Vanadium in Kelvin is 2183 K.
How Abundant is Vanadium?
Vanadium Atomic Radius
Abundant value of Vanadium is 120 mg/kg.
Atomic Number Of Vanadium
What is the State of Vanadium at Standard Temperature and Pressure (STP)?
State of Vanadium is Solid at standard temperature and pressure at 0℃ and one atmosphere pressure.
When was Vanadium Discovered?
Vanadium was discovered in 1801.
Atomic Number Of Vanadium